gastroenterology
Digestive Disease Week (DDW) 2011
Raising the Rate of Successful GERD Control: Focus on Improved Pharmacokinetics of Acid Suppressant Therapy
Chicago – Proton pump inhibitors (PPIs) are the standard for control of gastroesophageal reflux disease (GERD), but there is increasing appreciation for their limitations. Although PPIs were the first pharmacological agent with a high rate of success for healing esophagitis, a substantial minority of patients report incomplete control of symptoms on standard doses. This has generated a variety of strategies to improve symptom relief, most often a twice-daily regimen to replace the standard once-daily morning dose. The rationale for an extra dose of PPI is based on the physiology of acid pump production. Although PPIs irreversibly bind to acid pumps to prevent acid production, new pumps form constantly, particularly with meal stimulation. While the added dose of a PPI can bind to and inhibit the pumps that form subsequent to the first dose, it invites the potential for reduced compliance, defeating this potential advantage. A new approach is based on improving PPI pharmacokinetics.
Addressing the Need for New GERD Regimens
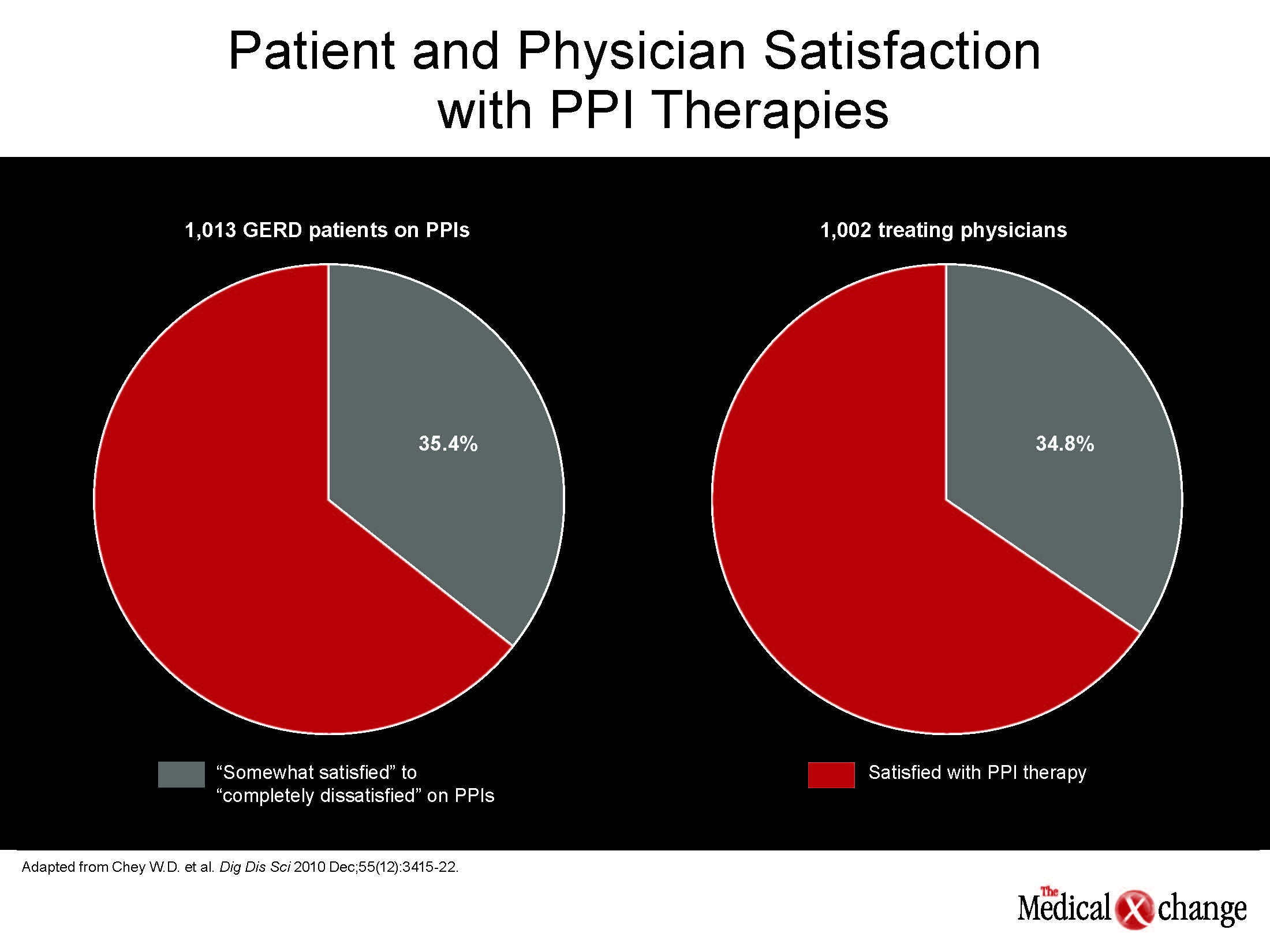
“The evidence that more than one-third of patients with GERD who are prescribed a PPI are not satisfied with therapy has generated a variety of efforts to improve care.”
The evidence that more than one-third of patients with gastroesophageal reflux disease (GERD) who are prescribed a proton pump inhibitor (PPI) are not satisfied with therapy has generated a variety of efforts to improve care (Fig. 1). These include more aggressive regimens with existing PPIs as well as a broad effort to develop new pharmacological options. While therapies with alternative mechanisms of action remain at experimental stages of development, simply improving the pharmacokinetics of PPI therapy is proving to be an immediate solution. The first agent to test and confirm the efficacy of this approach is dexlansoprazole, which has a dual delayed release pharmacology that substantially extends duration of action. This is important because the limitation of current therapies is not the efficacy of proton pump inhibition but the duration of effect.
“In the vast majority of GERD patients who are not controlled on standard PPI doses, the solution is typically a second dose before the evening meal rather than a higher dose. While there are data to suggest that more than 20% of GERD patients are now on BID [twice-daily] PPIs, these regimens are also associated with reduced adherence,” reported Dr. Onur Baser, Research Assistant Professor of Surgery, University of Michigan, Ann Arbor. He explained that this has been the key reason to pursue a PPI with more favourable pharmacokinetics. In presenting data from a new study to test this approach, Dr. Baser confirmed an adherence advantage for dexlansoprazole among BID PPI users even after they switched back to once-daily (QD) therapy.
In this study, employing real-world treatment data from a large pharmacy practice database, those GERD patients on BID PPI therapy were followed after they were switched back to dexlansoprazole or another QD therapy for any reason. These represented about 10% of the 32,718 patients in the database who were BID PPI users. Of those who switched, 601 individuals, or 2% of the BID population, began a prescription of dexlansoprazole, and 3,052, or 9.3%, switched to another PPI taken QD.
Reflecting expected practice patterns, those switched to dexlansoprazole had a greater degree of GERD severity as indicated by a history of Barrett’s (10.2% vs. 7%; P?0.005) or strictures and esophageal stenosis (6.9% vs. 4.3%; P?0.03) than those switched to another QD PPI. Prior to the switch, the dexlansoprazole patients also had a higher number of GERD-related office (93.5% vs. 86.4%; P?0.001) and outpatient clinic visits (54.7% vs. 40.2%; P?0.001). Moreover, they consumed more daily medication (1.69 vs. 1.59 daily doses; P?0.001).
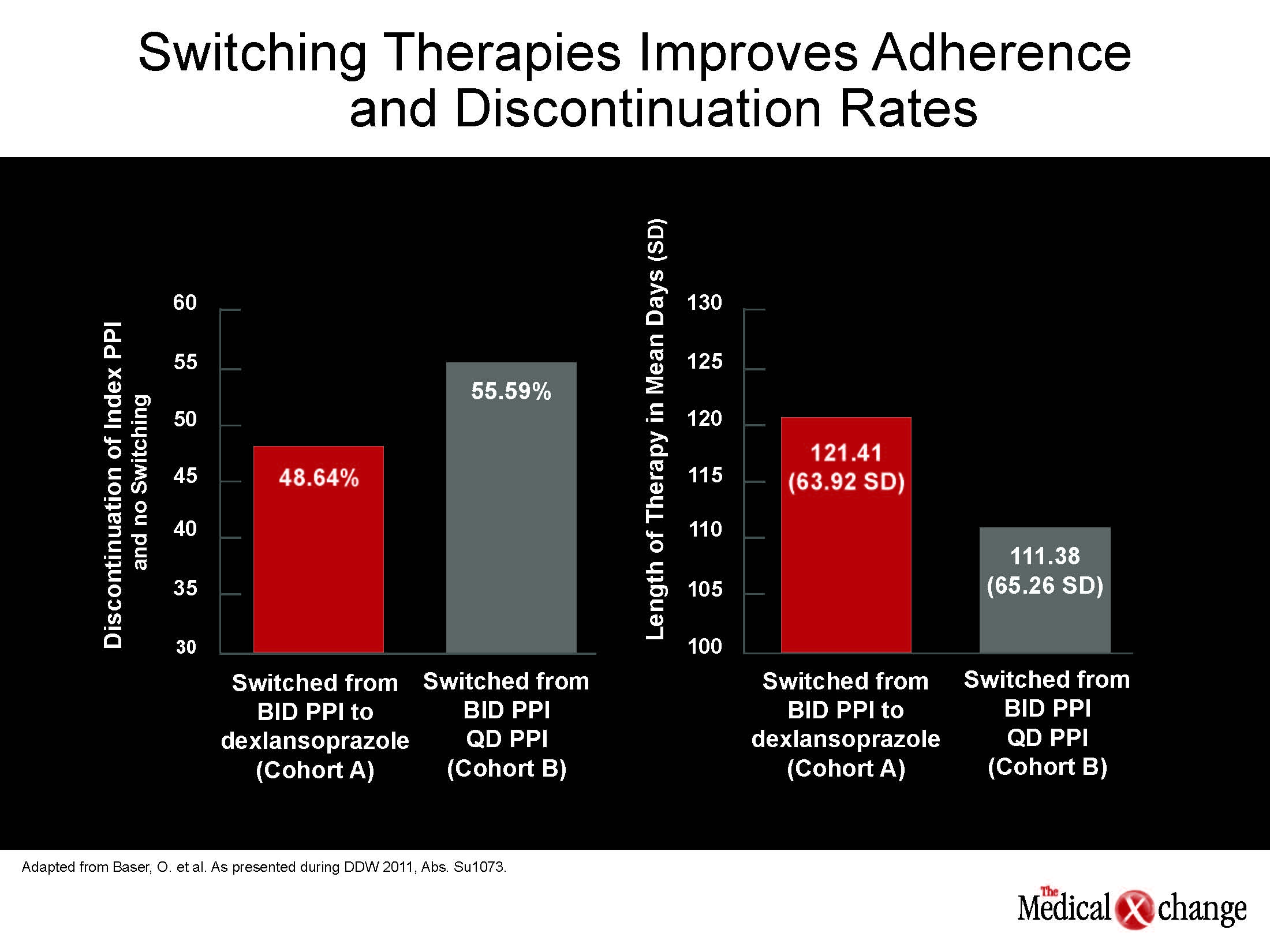
After the switch, discontinuation rates were lower among those switched to dexlansoprazole relative to another QD PPI (48.64% vs. 55.59%; P=0.0169), and patients remained on dexlansoprazole for a longer period (121.41 vs. 111.38 days; P=0.0078) (Fig. 2). Likely to reflect the greater satisfaction with therapy and fewer consultations, those on dexlansoprazole had similar overall and GERD-related health care resource costs despite higher anticipated pharmacy costs from treatment adherence. The overall results are consistent with more reliable efficacy stemming from improved pharmacokinetics.
“Overall, even though patients being switched to dexlansoprazole had more severe symptomatology, they were more likely to continue on therapy,” Dr. Baser reported. He indicated that the results of this large analysis are consistent with the expected activity of dexlansoprazole in the challenging population to which the pharmacology of the agent is particularly well suited.
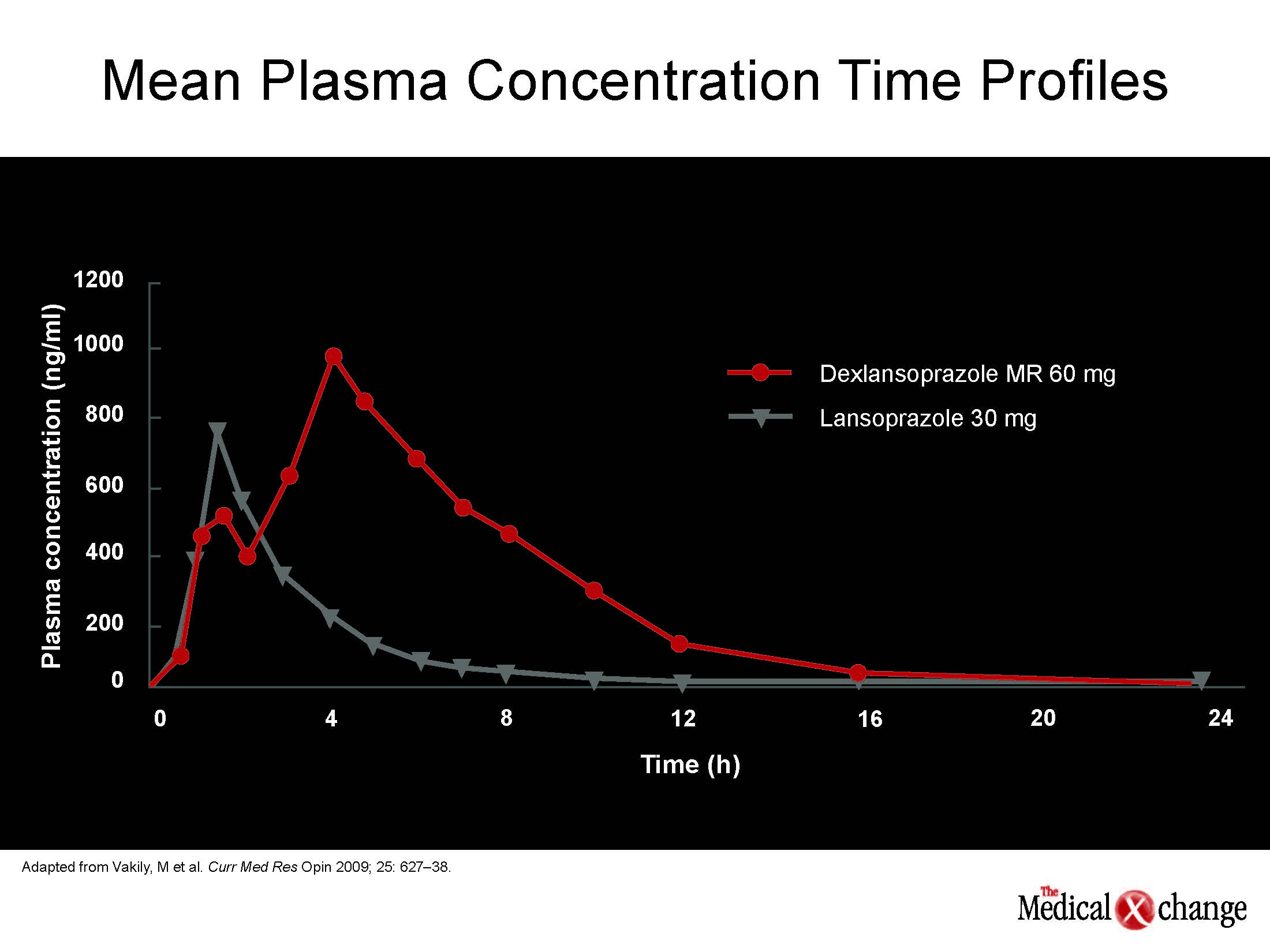
The PPI dexlansoprazole was created from the R-enantiomer of lansoprazole but it also incorporates two types of enteric-coated granules that extend release of active drug and provides two peak plasma levels. These dual peaks greatly improve the area-under-the-curve (AUC) exposure to the drug for each once-daily dosing period. The key advantage is that it maintains an active level of drug over a longer period than conventional PPIs, which typically have a half-life of less than two hours. In the context of the proton pump, which is the final pathway to gastric acid production and is more descriptively called the acid pump, this pharmacokinetics profile is highly relevant (Fig. 3).
Nocturnal heartburn is a particularly common problem in patients on a conventional once-daily PPI.
Specifically, PPIs bind irreversibly to acid pumps, which are activated by food. When properly taken about a half-hour before breakfast, conventional PPIs will shut down about 75% of the activated pumps, leaving just 25% of acid pumps still active. However, pumps are regenerated more or less constantly with reactivation of regenerated pumps with every meal. Over a 24-hour period, this regeneration restores another 25% of acid pump capacity, which is often sufficient to permit symptoms to recur. Nocturnal heartburn is a particularly common problem in patients on a conventional once-daily PPI.
“Dexlansoprazole is the first PPI with a dual delayed release formulation designed to prolong the plasma concentration-time profile and extend the duration of acid suppression while still permitting the PPI to be taken on a QD basis,” Dr. Baser explained. He indicated that this approach may help reduce the proportion of individuals who, even if their symptoms are improved, do not achieve satisfactory control of GERD symptoms on conventional PPIs. Those with nocturnal heartburn are particularly suitable candidates, but the greater duration of activity would be expected to be effective for any individual with residual symptoms.
Conclusion
A delayed-release PPI formulation has been developed to address the substantial proportion of individuals with GERD whose symptoms are insufficiently controlled or who have persistent esophagitis on a conventional PPI, including a conventional PPI taken twice daily. The once-daily delayed release agent preserves the convenience of a single daily dose while addressing the underlying physiology of failed or inadequate acid suppression. Pharmacy database analyses demonstrate that dexlansoprazole is already being employed in patients with greater GERD severity. In those on BID PPI who revert to a QD drug, dexlansoprazole was associated with significantly greater compliance, which may be a reflection of the greater satisfaction with symptom control.