cardiology
American Heart Association (AHA) Scientific Sessions 2015
PCSK9 Inhibitors Show Similar Efficacy in Diabetics as in Other High-Risk Groups
Orlando – PCSK9 inhibitors offer the same magnitude of low-density lipoprotein cholesterol (LDL-C) reduction in patients with diabetes mellitus as in other high-risk groups, according to a series of studies presented at the AHA Scientific Sessions. In this series, there was also no evidence that PCSK9 inhibition imposes any adverse influence on glucose metabolism. This latter finding is important in the context of new data that strengthen the modest but measurable association of statins with new-onset diabetes.
Appropriate Strategy in Diabetes
Studies presented at the AHA Scientific Sessions 2015 expand evidence that PCSK9 inhibitors are appropriate for bringing patients with diabetes mellitus to guideline-directed LDL-C goals. Although agents in this class will be used in most cases as an adjunct or substitute when statins are insufficient or poorly tolerated, PCSK9 inhibitors are not only more effective for lipid lowering than statins but do not share a counterproductive effect on glycemic control.
“We saw no association between the PCSK9 inhibitor and transition to impaired glucose control or diabetes mellitus.”
No Association with Incident Diabetes
“The data are reassuring,” reported Dr. Helen Colhoun, Clinical Professor, Diabetes Epidemiology Unit, University of Dundee, Scotland. Presenting an analysis of data collected from 10 Phase 3 trials with the PCSK9 inhibitor alirocumab, Dr. Colhoun said, “we saw no association between the PCSK9 inhibitor and transition to impaired glucose control or diabetes mellitus.” These findings contrast markedly with a statin study presented during the same session of oral papers. Characterized as the largest long-term study so far performed on the association between statins and impaired glucose metabolism, the adjusted hazard ratio (HR) for new-onset (incident) diabetes was 2.26 (95% CI 2.21 – 2.32; P<0.0001) for those exposed to statin relative to those who were not. “All statin types were associated with an increased incidence of diabetes without any statistically significant differences between them,” reported Dr. Tariq Ali Ahmad, Penn State Milton Hershey Medical Center, Pennsylvania. The data were derived from a nationally representative US database that compared 264,127 patients exposed to statins to an equal number not exposed. Risk adjusted for confounding factors such as obesity, the association between statin exposure and incident diabetes increased with age. The greatest risk was observed in patients initiating statin therapy above the age of 50 years. Sensitivity analyses yielded consistent results.
Phase 3 Program Data Analyzed
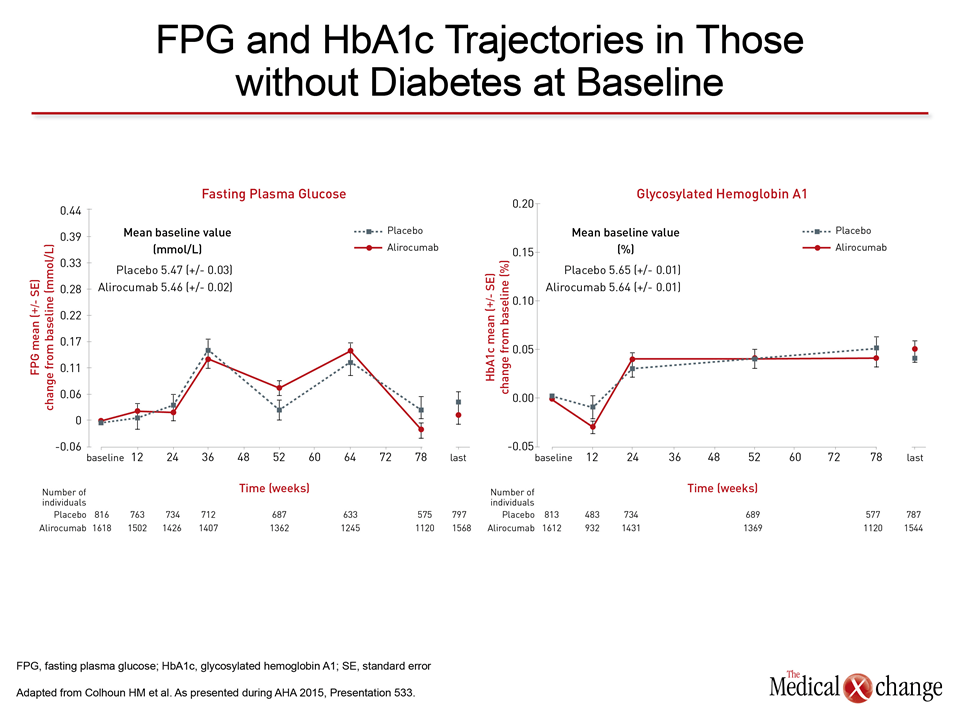
This association, reported previously, provided a rationale for a detailed evaluation of the experience with alirocumab in the ODYSSEY Phase 3 program, which has included ten Phase 3 trials that have enrolled 4,974 patients. In an analysis of fasting plasma glucose (FPG) and hemoglobin A1c (HbA1c) over time in the 3,448 patients without diabetes at entry, no differences were observed over time (Fig. 1). When data were divided between the placebo-controlled and the ezetimibe-controlled trials, the incidence of diabetes was consistently lower in patients randomized to alirocumab relative to placebo (1.7% vs. 2.6%) and to alirocumab relative to ezetimibe (1.5% vs. 2.1%). Furthermore, when evaluating the 1,969 patients in the ODYSSEY program with pre-diabetes at baseline, defined as FPG ≥ 5.5 but <6.9 mmol/L or HbA1c ≥5.7% but <6.5%, about 10% transitioned to diabetes over the course of follow-up, but there were no significant differences seen between treatment groups. In a separate analysis from the ODYSSEY Phase 3 program presented here, data from five Phase 3 studies confirmed that the lipid-lowering efficacy of alirocumab in the patients was at least as good in patients with diabetes as without. All were on maximally-tolerated statin therapy at entry. Efficacy from the studies was pooled for the two groups.
LDL-C Lowering Sustained
From baseline, the LDL-C reductions were slightly greater but not significantly different for the 1,048 patients with diabetes when compared to the 2,444 patients without (Fig. 2). The LDL-C reductions in both groups were sustained as long as patients remained on therapy, according to Dr. Henry N. Ginsberg, Associate Dean of Clinical and Translational Research, Columbia College of Physicians and Surgeons, New York City, NY. Consistent with the previously observed patterns, baseline levels of LDL-C were slightly lower (3 vs. 3.36 mmol/L) and triglycerides were somewhat higher (1.64 vs. 1.35 mmol/L) in those with diabetes when compared to those without. Modest improvements in lipid fractions other than LDL-C, such as Apo B, Lp(a), and high-density lipoprotein cholesterol (HDL-C) were observed “largely irrespective of baseline diabetes status,” Dr. Ginsberg reported.
Injection Site Reaction Rates Differ
Reported adverse events did not differ between alirocumab and placebo whether or not patients had diabetes. The exception was injection site reactions. Substantially fewer patients with diabetes complained of this event (approximately 3% vs. 7%). “The lower rate of injection site reactions reported by patients with diabetes is likely to be due to the fact that some of these patients take insulin and are more accustomed to self-injection,” Dr. Ginsberg speculated.
Conclusion
PCSK9 inhibition is as effective for LDL-C lowering in patients with diabetes as in those without. Further, this mechanism of action is not associated with the incident diabetes linked to statins. The findings encourage this lipid-lowering strategy in patients with diabetes unable to reach LDL-C treatment targets on traditional therapies.
Additional Slide
Figure 3 (Fig. 3).