cardiology
The 65th American College of Cardiology Annual Scientific Session & Expo (ACC 2016)
Increasing NT-proBNP Isolated as Surrogate for Adverse Outcomes in Heart Failure
Chicago – When N-terminal pro brain B-type natriuretic peptic (NT-proBNP) increases over a period of 6 months or more, the risk of heart failure hospitalization and all-cause death climbs with it, according to new data presented at the 2016 ACC. In placing these data in context, the authors suggested NT-proBNP is a powerful surrogate for outcome with potential application for monitoring patients and to judge treatment efficacy in clinical trials.
Slowing Heart Failure Progression
The prognostic importance of an elevated baseline NT-proBNP is already well recognized and was an entry criterion for the PARADIGM-HF trial, which also generated new data presented at the 2016 ACC meeting. In the latest set of data from that trial, a landmark study that associated a combination of the neprilysin inhibitor sacubitril and valsartan with an 20% reduction in the primary composite outcome of cardiovascular (CV) death or heart failure hospitalization relative to enalapril (McMurray et al. N Engl J Med 2014;371:993-1004), there was essentially no difference irrespective of background therapy. Both of the studies presented at the 2016 ACC meeting emphasize control of neurohormonal activation as key mechanism for slowing heart failure progression.
Reductions in NT-proBNP for Improved Prognosis
In the study of change in NT-proBNP, the change in the primary endpoint of all-cause mortality and heart failure hospitalization was compared in 363 patients in the Swedish Heart Failure Registry who showed a reduction in NT-proBNP at 7 months to the 291 who did not. At a median of 2.2 years of follow-up, those with improvement (reduction) in NT-proBNP had a 40% improvement (HR 0.602; P<0.001). For all-cause mortality, the reduction was 55% (HR 0.549; P=0.002). In this analysis, which was restricted to patients with a preserved ejection fraction, defined as >40%, “reductions in NT-proBNP levels over time are independently associated with an improved prognosis in heart failure patients,” confirmed Dr. Gianluigi Savarese, Department of Medicine, Karolinska Institutet, Stockholm, Sweden.
Reductions in NT-proBNP levels over time are independently associated with an improved prognosis in heart failure patients.
PARADIGM-HF: New Data
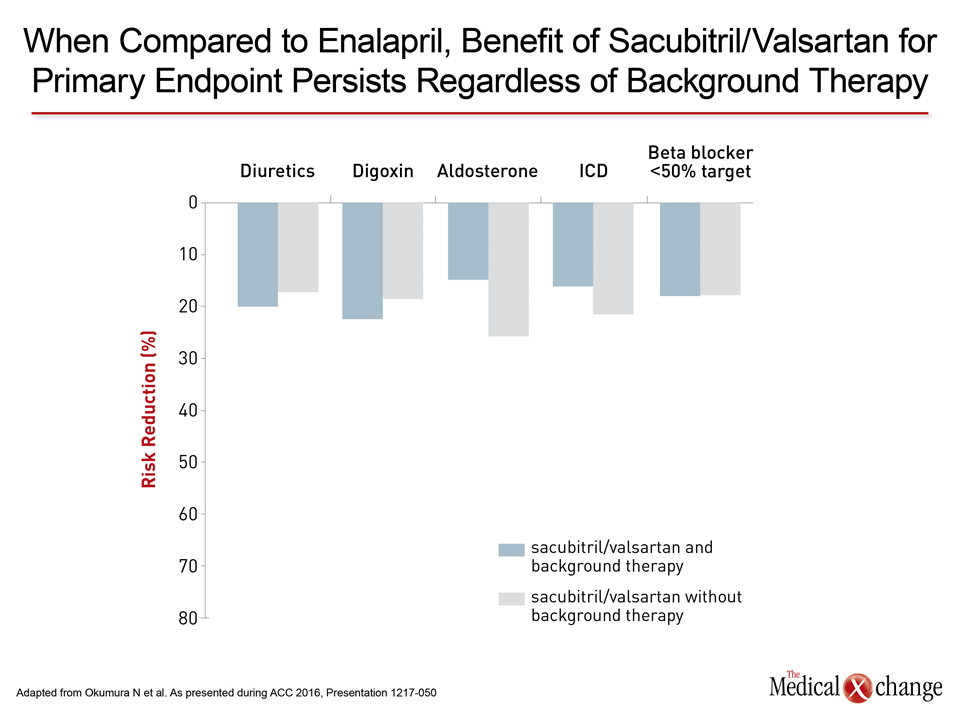
In the new data from PARADIGM-HF, hazard ratios for treatment with sacubitril/valsartan relative to enalapril were calculated for the primary composite endpoint across all baseline treatments, such as diuretics, digoxin, aldosterone and other mineralocorticoid receptor antagonists (MRA), implantable cardioverter defibrillator (ICD) and other implantable devices, and prior coronary revascularization (Fig. 1). While essentially all patients were on a beta-blocker at baseline, the relative benefit for this analysis was calculated for those at 50% or greater of the target dose versus those below.
Conclusion
“The advantages of sacubitril/valsartan relative to enalapril were incremental to any therapy offered to these heart failure patients,” reported Dr. John V. McMurray, Cardiovascular Research Centre, Glasgow, Scotland. He further noted that in this study, which required a NT-proBNP level of ≥600 pg/mL for entry, that the risk of CV death when evaluated separately was also proportionally reduced by about 20% for sacubitril/valsartan relative to enalapril across all background treatment subgroups.