Cardiology
European Society of Cardiology (ESC) Congress 2017
CANTOS Trial Advances Inflammatory Hypothesis of Coronary Artery Disease
Barcelona – In patients with a previous myocardial infarction (MI), a therapy targeted at an inflammatory cytokine has been associated with a significant reduction in major cardiovascular (CV) events. This clinical benefit, demonstrated in the placebo-controlled multinational CANTOS trial, which randomized more than 10,000 patients, confirms that inflammation is an important driver of coronary artery disease, a result that alters fundamental conceptions of disease pathology and establishes a new option for treatment.
Anti-inflammatory Therapy for CV Protection
“These findings represent the end game of more than two decades of research, stemming from a critical observation that half of heart attacks occur in people who do not have high cholesterol,” said CANTOS principal investigator Dr. Paul M. Ridker, Director, Center for Cardiovascular Disease Prevention, Brigham and Women’s Hospital, Boston, Massachusetts. The CANTOS data were presented as a latebreaker at this year’s ESC and simultaneously published in the New England Journal of Medicine (Ridker PM et al. N Engl J Med 2017; Aug 17 epub ahead of print).
In CANTOS, 10,061 patients with a previous MI and a high-sensitivity C-reactive protein (hs-CRP) of ≥ 2 mg/L were randomized to one of three doses of the monoclonal antibody canakinumab or placebo. All therapies were administered subcutaneously every three months. Canakinumab, which is approved in many countries for the treatment of rheumatologic disorders, targets IL-1β, a cytokine considered central to inflammation. At entry, 97% of patients were on statins and the median LDL-C was approximately 2.0 mmol/L.
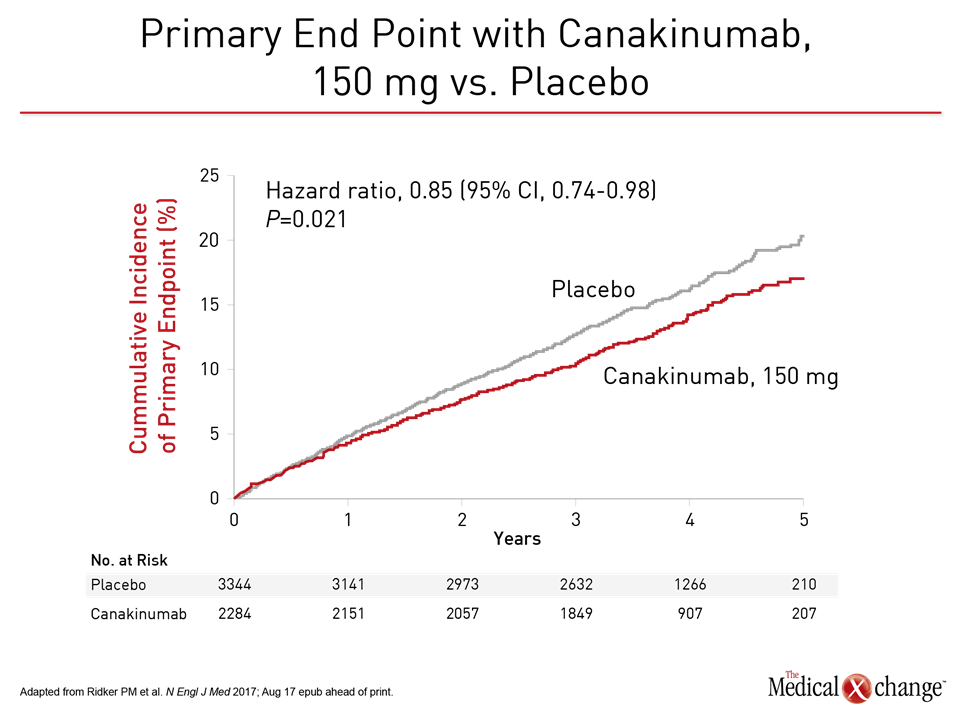
At a median follow-up of 3.7 years, the hazard ratio (HR) for the composite primary efficacy endpoint of nonfatal MI, nonfatal stroke, or CV death met the prespecified multiplicity-adjusted threshold for statistical significance for canakinumab 150 mg relative to placebo (HR 0.85; P=0.021) (Fig. 1). This 15% reduction in events was similar to that achieved with the highest dose of 300 mg (HR 0.86; P=0.031). The relative 7% reduction in events for the lowest dose (50 mg) did not reach statistical significance (P=0.3). For the 150 mg dose, there was also a significant 17% reduction in a secondary composite endpoint that included hospitalization for unstable angina leading to revascularization (P=0.005). At 48 months, there were significant reductions in hs-CRP for canakinumab relative to placebo in a dose relationship, reaching 41% for the highest dose.
As has been observed with other agents targeted at inflammatory cytokines, canakinumab was associated with a higher incidence of infections than placebo, including fatal infections (0.31 vs. 0.18; P=0.02). However, canakinumab was associated with a lower incidence of fatal cancers (0.45 vs. 0.64; P=0.02) in a dose-dependent manner. The agent was also associated with significantly lower rates of arthritis, gout, and osteoarthritis. There was no significant difference between canakinumab and placebo for all-cause mortality.
The results of CANTOS establish a new chapter in preventive cardiology, according to Dr. Ridker. He characterized the protection from CV events associated with diet and exercise as the first era and the protection from cholesterol lowering as the second era. The data from CANTOS, associating anti-inflammatory therapy with CV protection, “is cracking the door open on the third era.”
Conclusion
In a New England Journal of Medicine accompanying editorial, Dr. Robert Harrington, Chairman, Department of Medicine, Stanford University, California, largely agreed. He wrote, “CANTOS has helped move the inflammatory hypothesis of coronary artery disease forward.” He remains circumspect about the routine use of canakinumab or other anti-inflammatory agents until more is known about efficacy, safety, and cost, but he conceded that these data have created “scientific and clinical excitement” about the potential of “a new mechanism of action to attack.”