Ophthalmology
American Academy of Ophthalmology (AAO) 2018
New Agents and Combinations Promise Improved Glaucoma Control
Chicago – New therapeutic agents or drug combinations are improving the options for control of open angle glaucoma, according to a series of studies presented or discussed at this year’s American Academy of Ophthalmology (AAO) scientific meeting. Drug therapies are the first-line approach for reducing intraocular pressure (IOP), preventing functional impairment while delaying or avoiding surgery, but the limitations of single agents, such as prostaglandin analogs (PGAs), are drawing attention to combination strategies as well as driving interest in new agents. Both strategies are moving forward and promise to generate an evolution in routine management.
Clinical Trials Underline the Need for Early and Aggressive Treatment of IOP
Based on a large body of evidence that early control of IOP in open angle glaucoma improves outcomes, there is growing interest in combination regimens and new agents with novel mechanisms as a means of rapid pressure reductions, according to discussions at the 2018 annual AAO meeting. Several recent studies suggest progress. In one, presented prior to the AAO but included in discussion of new options, a fixed-dose combination of the alpha agonist brimonidine and the carbonic anhydrase inhibitor brinzolamide yielded impressive IOP reductions when combined with a PGA.
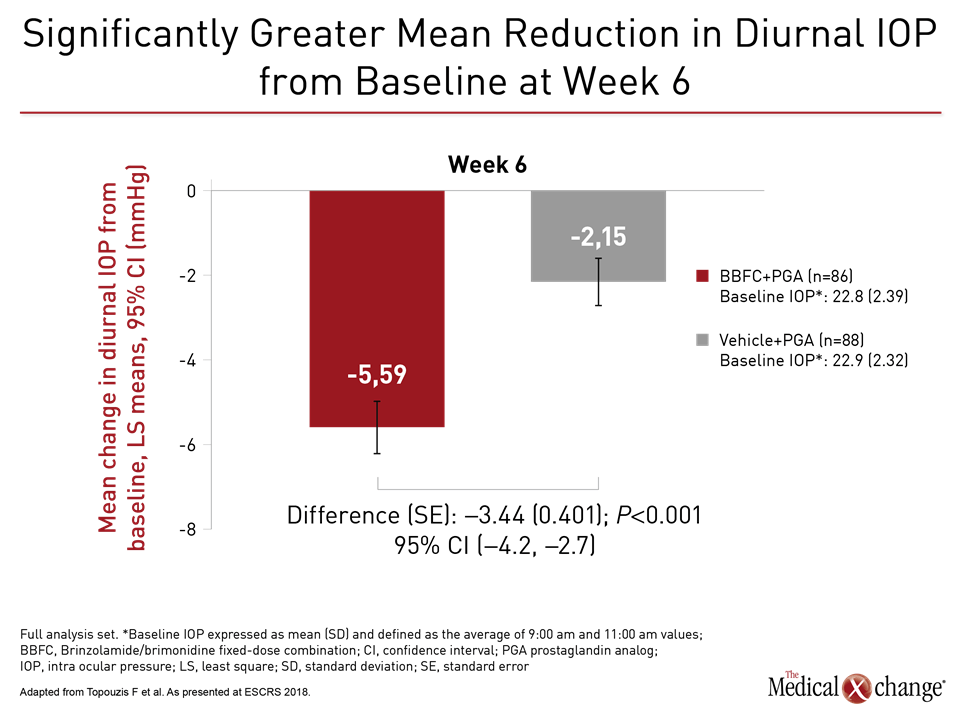
In this study, validating a combination that is already offered in separate dosing by many ophthalmologists in Canada and elsewhere, 187 open angle glaucoma patients with inadequately controlled IOP on PGA alone were randomized to receive a fixed-dose combination of brinzolamide/brimonidine or vehicle as an adjunctive treatment to PGA. The primary endpoint was change in diurnal IOP.
Results were impressive. At the end of the six-week trial, IOP pressure reductions from baseline were more than double for the three-drug combination (-5.59 vs. 2.15 mmHg; P<0.001), according to the principal investigator, Dr. Fotis Topouzis, Department of Ophthalmology, Aristotle University, Thessaloniki, Greece (Fig. 1). Based on a favourable safety profile, Dr. Topouzis, who presented these data at the European Society of Cataract and Refractive Surgeons (ESCRS) just before the 2018 AAO, called this strategy “a suitable treatment option” for those with persistent IOP on single agents.
In those randomized to the combination arm, the fixed-dose of brinzolamide 1%/brimonidine 0.2% was taken twice daily with once-daily PGA. The randomization to the study arms was conducted after a 28-day run-in/wash-out period followed by a 3- to 8-day eligibility screen. At baseline, the mean diurnal IOP was approximately 23 mmHg. The PGA taken at baseline was latanoprost in 40% of patients, bimatoprost in 34% of patients, and travoprost in 26% of patients.
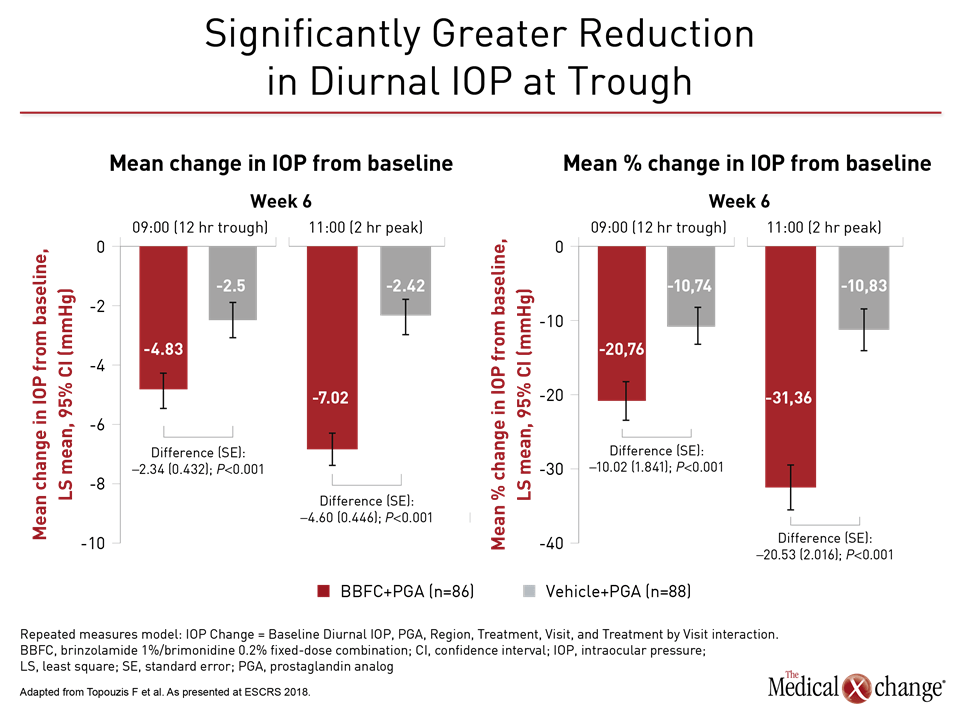
When combined with a PGA, the advantage of the fixed-dose combination over vehicle alone was equally robust for the secondary endpoints (Fig. 2)). Of ocular adverse events, hyperemia was more common on the combination (5.3% vs. 1.1%). Blurred vision was the most common adverse event in the arm on PGA plus vehicle, but the rates were similar to the combination arm (2.2% vs. 2.1%). Overall, Dr. Topouzis called adverse events “consistent with the known safety profiles of brinzolamide, brimonidine and PGA.
Commonly, treatment for IOP in patients with open angle glaucoma has been performed with a stepped-therapy approach starting with a PGA and then switching among alternative monotherapies before moving to combination treatment, but many believe a more aggressive approach is warranted. The evidence that prompt control of elevated IOP improves outcomes dates back more than a decade. In the landmark Early Manifest Glaucoma Trial, for example, immediate lowering of IOP with either surgery or pharmacologic therapy significantly reduced progression at six years irrespective of age, degree of IOP, or visual acuity at baseline (Heijl A, et al; Arch Ophthalmol. 2002;120:1268-79).
More Medical Options for IOP Moving Forward
Several new options have received recent approval from the US Food and Drug Administration and may be soon available in Canada. These include latanoprostene bunod ophthalmic solution 0.024%, which is a nitric-oxide donating PGF2 alpha analog, and netarsudil ophthalmic solution 0.02%, which is a Rho kinase inhibitor. Both are once-daily therapies. At this year’s AAO, Dr. Paul Harasymowycz, Chief of Glaucoma, University of Montreal and Medical Director, Bellevue Ophthalmology Clinics and the Montreal Glaucoma Institute, Quebec, said that these alone or in novel combinations are promising.
“It could be that a patient is on a PGA and a fixed-dose combination and we add netarsudil 0.02%,” Dr. Harasymowycz said. He explained that unlike PGA agents or alpha agonists, which reduce IOP by increasing uveoscleral outflow, or brinzolamide, which reduces aqueous humor production, netarsudil reduces IOP by increasing aqueous humor outflow through the trabecular meshwork route.
Due to the unique mechanism of action, “we expect our patients to get additional IOP lowering in addition to their current drops,” Dr. Harasymowycz reported. However, he cautioned that rates of conjunctival hyperemia have been high with netarsudil.
Based on studies that suggest the lower the IOP the better for preventing progression of glaucoma, Dr. Harasymowycz said that even modest improvements achieved by adding drugs should be considered meaningful when these combinations are well tolerated.
He called the incremental improvements in IOP reductions with combinations “exciting.” According to Dr. Harasymowycz, several of the combinations in clinical testing show incremental reductions in IOP with low or no additional burden of adverse events.
Conclusion
In the context of evidence that early and aggressive control of IOP in patients with open angle glaucoma leads to better outcomes, studies showing meaningful pressure reductions with combinations of existing or emerging agents promises better long-term outcomes. In this regard, fixed-dose combinations may be of particular value. By reducing the number of drops required to achieve control of IOP, this approach has the potential to keep the burden of therapy low to facilitate compliance. With combinations known to increase the likelihood of IOP control, maximum compliance is the common pathway to clinical gains.