Cardiology
European Society of Cardiology (ESC) Congress 2019
For Stroke Prevention in Patients with Atrial Fibrillation Undergoing PCI, Double Antiplatelet Therapy Found to be Safe
Paris – In patients with atrial fibrillation (AF) who had a percutaneous coronary intervention (PCI), a non-vitamin K antagonist oral anticoagulant (NOAC) plus a P2Y12 inhibitor has emerged as a new option for preventing ischemic events. In the phase 3b study, called ENTRUST-AF PCI, a full dose dual therapy was associated with a numerically lower risk of bleeding when compared to a triple therapy anchored with a vitamin K antagonist (VKA). It was as effective for preventing a composite of key adverse events, including death from cardiovascular (CV) causes, stroke, and stent thrombosis.
The ENTRUST-AF PCI study is the fourth multicenter trial with a NOAC for this group of patients, but it is the first to be conducted with a full-dose, once-daily NOAC-based combination regimen showing an acceptable balance of bleeding risk and ischemic-event protection. The study was conducted with edoxaban, the most recently approved factor Xa inhibitor. ENGAGE AF-TIMI 48, a previous multinational study, already established the efficacy and safety of edoxaban for stroke prevention in AF patients.
On the basis of the newly-completed ENTRUST-AF PCI study, “edoxaban 60 mg once daily plus a P2Y12 inhibitor can be considered non-inferior to a triple therapy with a vitamin K antagonist,” reported principal investigator Dr. Andreas Goette, Head, Department of Cardiology and Intensive Care Medicine, St. Vincenz Hospital, Paderborn, Germany.
NOACs Increasingly Preferred for AF Risk
This finding is important because NOACs are increasingly preferred over a VKA for their convenience and more predictable activity. However, one of the four prior studies was conducted with dabigatran, the only available direct thrombin inhibitor, which requires twice-daily dosing. Another was conducted with apixaban, administered in a full dose, twice daily, and the last studied rivaroxaban once daily, but via a low-dose strategy.
The ENTRUST-AF PCI study is the latest to attempt to identify “the right combination of drugs at the right doses to provide the greatest prevention against ischemic events with the least risk of increased bleeding,” explained the ESC-invited discussant, Dr. Renato Lopez, Professor of Cardiology, Duke University School of Medicine, Durham, North Carolina. Based on ENTRUST-AF PCI, a NOAC plus a P2Y12 inhibitor without aspirin “should now be considered the preferred regimen.”
ENTRUST-AF PCI tested “the right combination of drugs at the right doses to provide the greatest prevention against ischemic events with the least risk of increased bleeding.”
In ENTRUST-AF PCI, which involved 186 participating centers in 18 countries, 1506 patients with AF who were indicated for anticoagulation following PCI were randomized to one of two anticoagulation strategies. Those in a dual therapy treatment arm received 60 mg once daily of edoxaban plus a P2Y12 inhibitor. Those in a triple therapy treatment arm received VKA in combination with a P2Y12 inhibitor plus 100 mg of daily aspirin. The primary outcome was a composite of major or other clinically relevant non-major bleeding events. The regimens were started four hours to five days after undergoing PCI (median 45.1 hours) and continued for 12 months.
Annualized Bleeding Rate
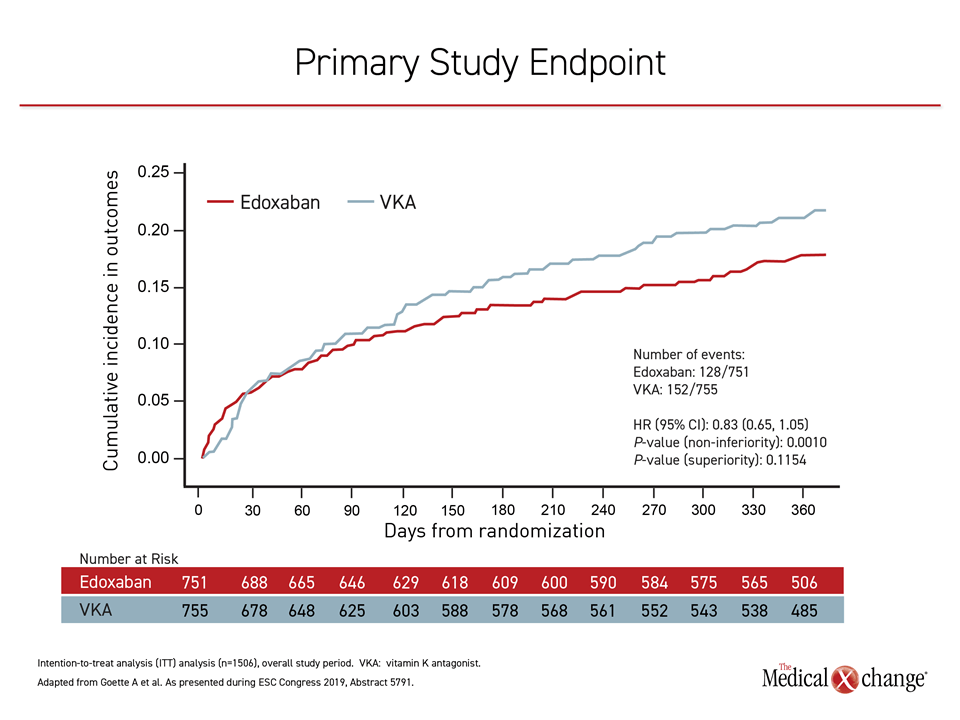
At the end of one year, the annualized bleeding event rate was 20.7% in the edoxaban-based arm versus 25.6% in the VKA-based arm. The outcome was statistically significant for demonstrating non-inferiority (P=0.001). For superiority, which was also being explored as a study hypothesis, the numerical advantage, which was persistent after the first two weeks for the length of the trial, fell short of statistical significance (P=0.115) (Figure 1).
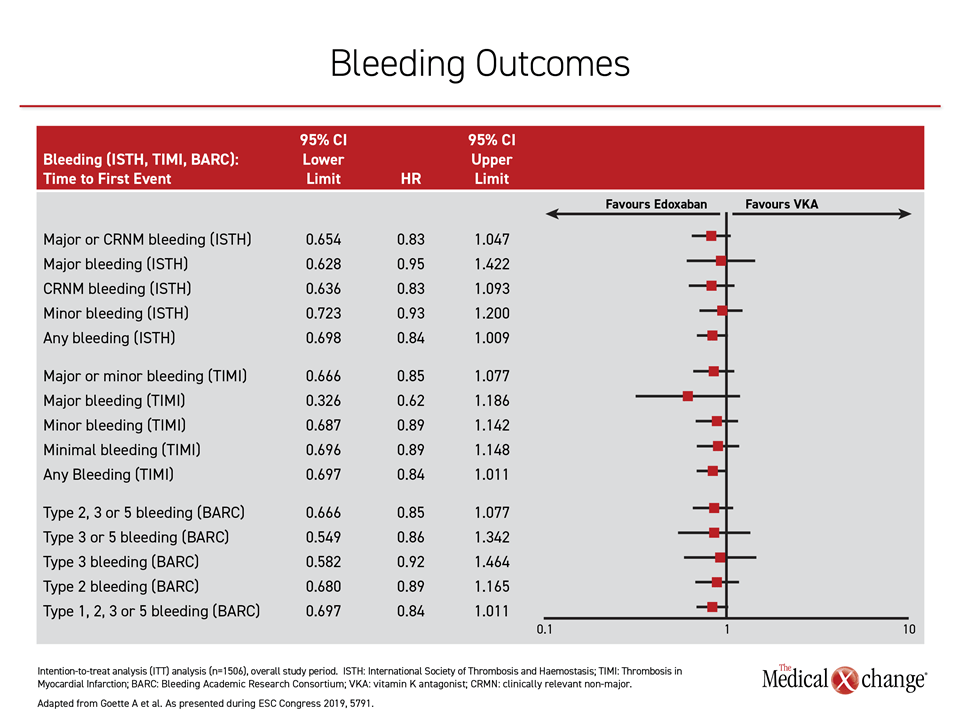
“We looked at multiple definitions of bleeding, which consistently favored edoxaban numerically at the end of the study on intention-to-treat analysis,” Dr. Goette reported (Figure 2).
“We looked at multiple definitions of bleeding, which consistently favored edoxaban numerically at the end of the study on intention-to-treat analysis.”
Similar Ischemic Event Protection
For the efficacy outcome, which was a composite of death from CV causes, stroke, systemic embolic events, myocardial infarction (MI) or definite stent thrombosis, rates in the two arms were similar, according to Dr. Goette. The ENTRUST-AF PCI results were published simultaneously with their presentation at the ESC (Vranckx P et al. Lancet 2019; September 3, epub ahead of print).
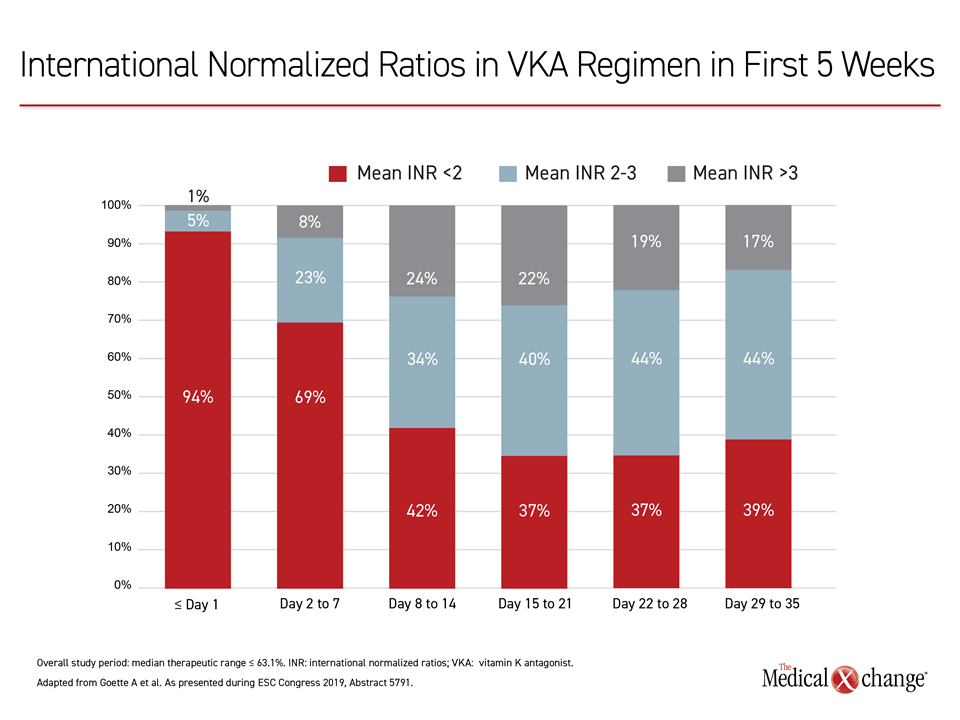
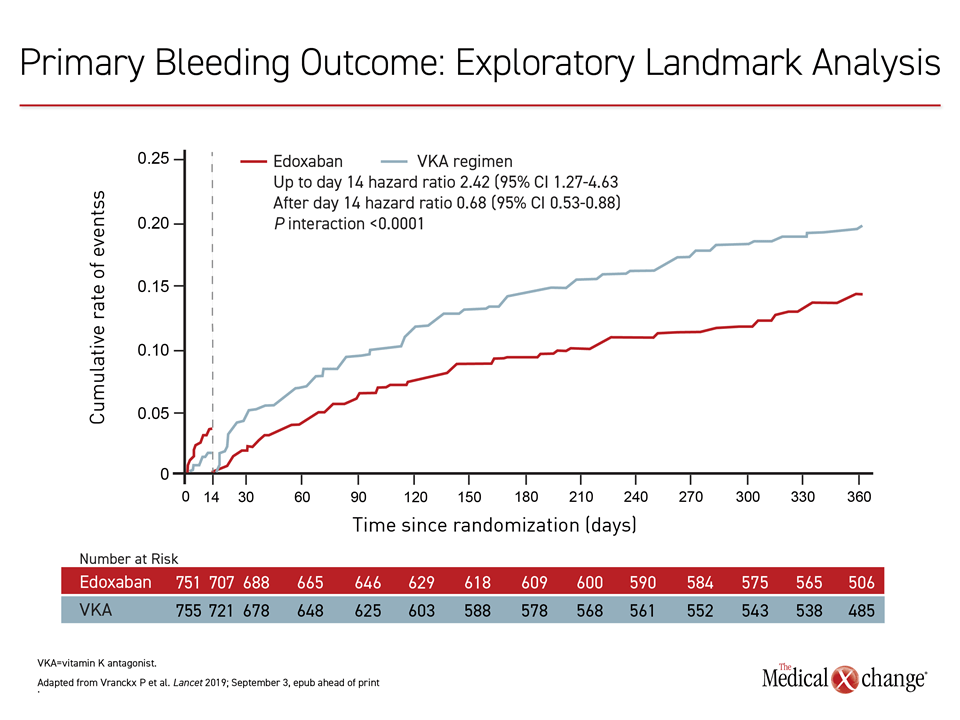
In the first two weeks of the study, there was a non-significant lower bleeding rate for the VKA-based regimen relative to the edoxaban-based arm, but this was reversed after that point. In a post-hoc analysis to explore this difference, the doses of the VKA were found subtherapeutic after this timeframe on the basis of the international normalized ratio (INR). Nearly 70% of VKA patients had subtherapeutic INRs on days 2-7.
Most Patients on Once-daily Dosing
In ENTRUST-AF PCI, although the dual therapy treatment arm received 60 mg once daily of edoxaban plus a P2Y12 inhibitor, the study permitted a 30 mg once-daily dose of edoxaban in patients with low creatine clearance (≤50 mL/min), bodyweight (≤60 kg), or concomitant use of specified potent P-glycoprotein inhibitors. Most patients in both arms (92.5%) also received clopidogrel by default, however, prasugrel or ticagrelor could also be used at the investigator’s discretion. By avoiding ticagrelor, which requires twice-daily dosing, most patients in both arms were maintained on a once-daily regimen.
Rivaroxaban, another once-daily NOAC also compared favorably for bleeding to a VKA-based therapy in the PIONEER-AF PCI trial (Gibson CM et al. N Engl J Med 2016;375:2423-2434), but rivaroxaban, unlike edoxaban in ENTRUST-AF PCI, was tested in two reduced-dose strategies.
However, the ENTRUST-AF PCI study results are consistent with other studies of NOACs in this study population. According to Dr. Lopez, the data confirm that “triple therapy with a VKA, P2Y12 inhibitor, and aspirin should be avoided.” Overall, the advantage of NOACs to VKAs is that they eliminate the need for systematic monitoring and dose adjustment to maintain therapeutic anticoagulation.
Conclusion
In AF patients requiring anticoagulation after PCI, the ENTRUST-AF PCI trial demonstrated that full-dose edoxaban plus a P2Y12 inhibitor provides a persistently lower risk of bleeding than a VKA-based regimen. It was non-inferior for this endpoint while providing similar protection against a composite of ischemic events. The data supports this strategy as a preferred option over VKA-based therapy.