Expert Review
The Aging HIV Patient
Chapter 2: Cardiovascular Disease
Dominique Costagliola, PhD
Université Pierre et Marie Curie, Paris, France
Early death from cardiovascular (CV) complications in aging individuals infected with HIV is a growing challenge in chronic HIV management. There are numerous factors that have converged to increase CV risk in individuals infected with HIV compared to age-matched individuals without infection. These include a greater prevalence of CV risk factors, the adverse effects of several antiretroviral agents that appear to contribute to CV risk, and a direct or indirect effect of HIV itself. In HIV patients with otherwise well controlled viremia, CV disease is expected to become an increasingly important cause of preventable death. The effort to modify the impact of this potential crisis depends on heightened awareness of the risks in HIV patients and aggressive monitoring and intervention for modifiable risk factors. Individualization of therapy will be increasingly important when attempting to reduce CV risks in the context of sustained HIV suppression.
Background
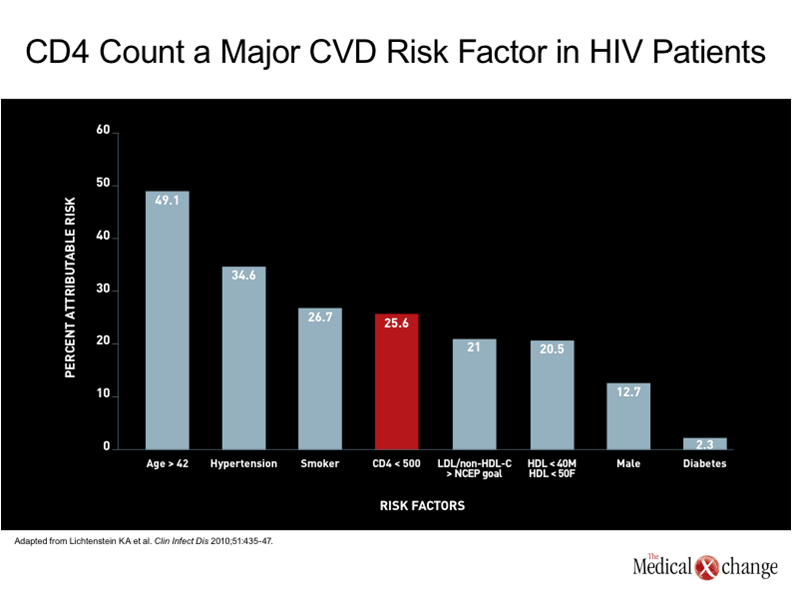
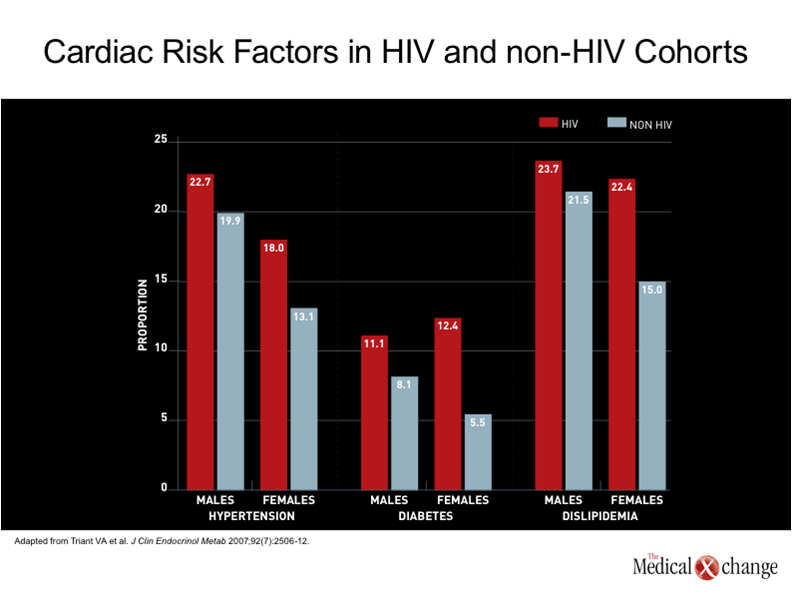
The introduction of combined antiretroviral therapy (cART) was associated with a large reduction in HIV-related deaths, but had a very modest influence on the rate of death from cardiovascular (CV) complications. (1)Prior to cART, dilated cardiomyopathy secondary to infective endocarditis was a major source of cardiovascular complications from HIV.(2)Since the introduction of cART, infective endocarditis remains an important source of adverse events, including death, in patients on cART who continue to use illicit intravenous drugs,(3)but suppression of HIV has reduced the risk for the opportunistic infections associated with this complication.(4)Although other types of CV complications were observed in patients with HIV prior to the introduction of cART, such as cardiac Kaposi’s sarcoma,(5)CV disease was not a major cause of death because of the greater importance of other risks.(6) The potential for CV disease to emerge as a significant cause of death in aging patients with well controlled HIV can be predicted on the simple basis that CV disease is the most important cause of death worldwide overall, particularly in aging individuals.(7)In Canada, vascular diseases account for nearly one third of all deaths and cause only slightly fewer deaths than all malignancies combined.(8)In a recent analysis of attributable risk for CV disease among a large cohort of HIV patients, a low CD4 count produced the fourth greatest influence. Age greater than 42 years provided the greatest.(9) (Fig. 1). Moreover, additional factors may exacerbate the effect of age. Evidence that CV disease might be a particular problem in an HIV population on cART therapy was first identified very early after protease inhibitors (PIs) were introduced into the routine management of this infection.(10)Subsequent studies reinforced the specific risk posed by PIs by showing increasing risk of myocardial infarction (MI) with increasing exposure to PIs.(11)The suspected mechanisms included disturbances in serum lipid levels and insulin metabolism,(12)but other effects, such as elevated fibrinogen levels,(13)have also been identified as potential contributors to this risk. One study calculated a 29% increase in the age-adjusted risk of MI for each year of HAART exposure.(14) However, the increased risk of CV disease cannot be wholly attributed to PI exposure or even to antiretroviral therapy overall. Rather, numerous cohort studies have associated HIV infection itself with an increased risk of CV disease relative to rates in non-infected individuals,(9, 15)including studies that found an increased risk after adjusting for hypertension, diabetes, and dyslipidemia (16) (Fig. 2). In a study which compared health claims data in two large hospitals in the United States, the MI rate was almost twice as great in those with HIV as in those without HIV after multiple adjustments, including age (11.13 vs. 6.98 per 1000 patients).(16) It is important to evaluate the contributors to the increased risk of CV disease in HIV patients in order to develop meaningful strategies of treatment, but the demographic shift that is increasing the proportion of HIV-infected individuals in age ranges where CV and other chronic diseases are common are making this an inevitable and increasing challenge in care of the individual with HIV.(17)Although many of the risk factors for CV disease, such as dyslipidemia, diabetes, hypertension, and renal impairment, are progressive conditions that generate an increasing and cumulative risk for MI and other CV diseases over time, the current evidence indicates that CV events will begin at a substantially earlier age in those with HIV than in those who are not infected.(18-19)
Pathogenesis
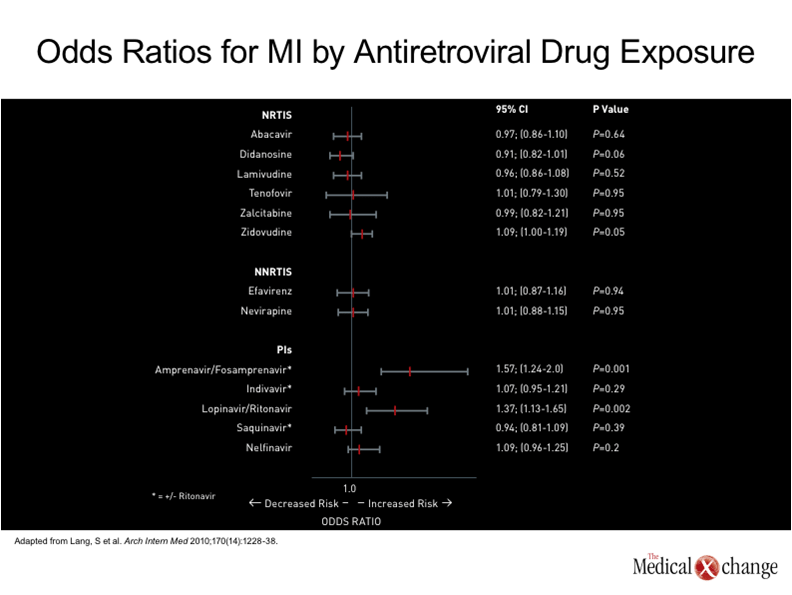
The accelerated aging phenotype that includes earlier expression of cardiovascular disease is not fully understood, but there are numerous and increasingly well supported theories which indicate that systemic immune activation plays a role. While the concept originally centered on immune function alone, producing the term immunosenescence, the effects of an upregulated immune system has been observed in all aging individuals.(20)However, this process appears to begin at a much younger age in individuals infected with HIV, and the accelerated senescence clearly extends to all or most physiologic systems.(21)The link between aging of the immune system and such organs as the heart could be an inability to suppress inflammation, which is known to contribute to atherosclerosis,(22)but other processes, such as shortening of telomeres on myocytes or other cells important to cardiovascular health, may also be involved.(23) In many patients, including those with a familial susceptibility, cardiovascular disease is the culmination of pathogenic processes driven by numerous risk factors, such as hypertension, diabetes, hyperlipidemia, renal impairment, and smoking (24) (Fig. 3). Several cohort studies have found that all or most of these risk factors are more common in an HIV-infected population.(15, 25)However, the acceleration of cardiovascular disease is greater than that predicted by these risk factors alone. In a study that evaluated subclinical coronary atherosclerosis by measuring coronary artery calcium (CAC) deposits with computed tomography (CT), a validated tool for evaluating CV disease progression, an increase in coronary age based on findings was observed in 69% of 233 HIV infected adults.(26)Even though HIV was well controlled in these individuals, who had a median age of 43 years, the risk of increased coronary age was associated with increasing duration of HIV infection, increasing use of antiretroviral therapies, and low CD4+ cell count nadir, along with traditional risk factors. Another study using CAC have also associated HIV with a more advanced vascular age than anticipated in non-HIV infected individuals.(27) The ability of aging processes in other organs to advance cardiovascular disease should not be overlooked. For example, diabetes mellitus, a major risk factor for CV disease, is also more prevalent in individuals with HIV than those who are not infected.(28)This spectrum of aging processes has produced what is being termed the HIV-related frailty syndrome.(29)While this deserves its own attention, these aging processes are relevant to the effort to address CV disease as a specific endpoint of this phenomenon. The specific contribution of antiretroviral therapies to CV risk may be relatively modest in the context of the acceleration in CV disease and the accompanying risk factors. The initial cohort studies that associated HAART with increased CV risk primarily implicated PI therapy,(30)leading to the theory that this risk was secondary to altered lipid or insulin metabolism caused by this class of antiretroviral agent.(31)However, this association was largely made prior to appreciation of the role of the infection itself in promoting CV disease. Moreover, many of the newer PIs, such as atazanavir, have not been associated with abnormalities in lipid metabolism.(32)The reduced risk of lipid abnormalities with newer PIs may minimize the CV risk, but other pro-thrombotic effects, such as elevated levels of fibrinogen, have been associated with agents in this class.(13) The D:A:D cohort found an association between abacavir and didanosine exposures and the risk of myocardial infarction but not stroke.(33)As abacavir was and remains a commonly used NRTI, several other groups attempted to reproduce this specific association with inconsistent results.(34)The potential problem of observational studies is limiting the bias and effective adjustment for confounders, such as preferential use of one NRTI over another in individuals with specific risks relevant to CV disease, such as renal impairment. Several studies have been unable to associate abacavir with any changes in vascular function consistent with CV risk, such as altered endothelial function, increased inflammation, or increased coagulation,(35-36)and the clinical significance of this association, if any, remains unclear. In nested case control study employing data from the French Hospital Database on HIV, the only antiretroviral agents that significantly increased the odds ratio of MI were PIs (19) (Fig. 4) The effort to prevent or modify the progression of CV disease in aging individuals with HIV is an appropriate target of efforts to prolong life in this population, but this risk must be considered in context. In particular, there may be no goal more important for survival than effective and sustained suppression of HIV. Moreover, individual patients may have more imminent health problems, such as renal impairment or liver disease, which may be more urgent than CV risk management. However, CV disease among the most frequent causes of death independent of HIV disease in most industrialized countries, including Canada.(37)In those individuals with HIV, the risk of CV disease appears to be magnified.
Diagnosis
Cardiovascular risk is not evenly distributed even within the HIV population, making individual risk assessments essential. Risk factors for CV disease should be considered even in relatively young individuals because of the opportunity early diagnosis offers for early intervention and improved outcome. The modifiable risk factors are the same as those observed in a non-HIV population, including smoking, hypertension, hyperlipidemia, hyperinsulinemia, and renal impairment. However, the age-related risk factors, such as hypertension, are likely to emerge at a younger age in those with HIV. As a result, tighter monitoring and more aggressive control is appropriate. There are no guidelines for early diagnosis of CV disease in HIV patients, but it is reasonable to adapt guidelines recommended for individuals without HIV because the contributing risk factors are similar. For asymptomatic individuals, several risk scoring systems are available, including the Framingham,(38)SCORE,(39)and Reynolds methods.(40)In general, these advice initial assessment of familial risk as well as evaluation of standard risk factors, such as smoking, blood pressure, serum lipid levels, and fasting glucose levels. More sophisticated testing is not generally recommended in individuals with low or intermediate risk, defined as an estimated 10-year risk of a CV event of <5%. However, such tests as exercise electrocardiography (ECG) or echocardiography is recommended in patients with greater degrees of baseline risk even if asymptomatic. In recommendations from the American Heart Association (AHA), the role of such tests as ECG or echocardiography is generally reserved for individuals over the age of 50,(41)but 26% of HIV patients with a mean age of 41 years had diastolic dysfunction and 40% had left atrial enlargement in one prospective echocardiographic study,(42)suggesting that earlier assessments may be appropriate in this population.(43)
Management
The single most important goal in patients with HIV is to provide sustained suppression of viremia. This is valid even when considering how to manage CV risk, because uncontrolled HIV is likely to pose a far greater immediate threat to survival than CV disease even in advanced stages. In patients on PIs, for example, neither elevated lipids nor the presence of CV disease is an absolute contraindication even to those PIs that adversely affect lipids. In individuals with elevated serum cholesterol on a PI, statin therapies may be sufficient to reach target reductions. While switching therapies to modify CV risk is reasonable, it should not be performed when the risk includes the potential for loss of HIV suppression. CV risk factor management in patients with HIV should be directed by goals and treatments recommended in the non-HIV population. This includes the same targets for blood pressure, serum lipids, and blood glucose as recommended in general guidelines. Due to the greater risk of CV disease, efforts to control risk factors, including smoking, should be considered urgent even in relatively young patients. Although the acceleration in aging processes has now been postulated in numerous organ systems, the frequency with which CV events are life-threatening should be a particular motivation for clinicians and patients to address CV risks and to adhere to treatment recommendations.
Conclusion
The most important defense against CV disease in aging individuals with HIV is greater awareness. Evaluation of risk factors early in life, including the presence of familial susceptibility, will help outline strategies of prevention that may slow the process. While individuals with HIV appear to have more risks for CV disease, the biggest threat is the earlier onset of CV disease. In addition, increased risk of CV disease may be driven in part by other accelerated conditions of age-related pathogenic process, such as hypertension, diabetes, and renal dysfunction. Close monitoring and aggressive treatment of CV risk factors should be immediately incorporated into HIV care.
References
1. Bozzette SA, Ake CF, Tam HK, Chang SW, Louis TA. Cardiovascular and cerebrovascular events in patients treated for human immunodeficiency virus infection. N Engl J Med 2003;348(8):702-10. 2. Barbaro G, Di Lorenzo G, Grisorio B, Barbarini G. Incidence of dilated cardiomyopathy and detection of HIV in myocardial cells of HIV-positive patients. Gruppo Italiano per lo Studio Cardiologico dei Pazienti Affetti da AIDS. N Engl J Med 1998;339(16):1093-9. 3. Chaves AA, Mihm MJ, Basuray A, Baliga R, Ayers LW, Bauer JA. HIV/AIDS-related cardiovascular disease. Cardiovasc Toxicol 2004;4(3):229-42. 4. Bijl M, Dieleman JP, Simoons M, van der Ende ME. Low prevalence of cardiac abnormalities in an HIV-seropositive population on antiretroviral combination therapy. J Acquir Immune Defic Syndr 2001;27(3):318-20. 5. Barbaro G, Di Lorenzo G, Grisorio B, Barbarini G. Cardiac involvement in the acquired immunodeficiency syndrome: a multicenter clinical-pathological study. Gruppo Italiano per lo Studio Cardiologico dei pazienti affetti da AIDS Investigators. AIDS Res Hum Retroviruses 1998;14(12):1071-7. 6. HIV/AIDS Epidemiology Annual Report 2003. San Francisco Department of Public Health, 2003. (Accessed February 4, 2011, at www.dph.sf.ca.us/PHP/AIDSSurvUnit.htm.) 7. Cardiovascular Diseases (Accessed February 3, 2011, at http://www.who.int/mediacentre/factsheets/fs317/en/index.html.) 8. Age-standardized mortality rates by selected causes, by sex. 2011. (Accessed February 4, 2011, at http://www40.statcan.gc.ca/l01/cst01/health30a-eng.htm.) 9. Lichtenstein KA, Armon C, Buchacz K, et al. Low CD4+ T cell count is a risk factor for cardiovascular disease events in the HIV outpatient study. Clin Infect Dis 2010;51(4):435-47. 10. Gallet B, Pulik M, Genet P, Chedin P, Hiltgen M. Vascular complications associated with use of HIV protease inhibitors. Lancet 1998;351(9120):1958-9. 11. Mary-Krause M, Cotte L, Simon A, Partisani M, Costagliola D. Increased risk of myocardial infarction with duration of protease inhibitor therapy in HIV-infected men. AIDS 2003;17(17):2479-86. 12. Calza L, Manfredi R, Chiodo F. Dyslipidaemia associated with antiretroviral therapy in HIV-infected patients. J Antimicrob Chemother 2004;53(1):10-4. 13. Madden E, Lee G, Kotler DP, et al. Association of antiretroviral therapy with fibrinogen levels in HIV-infection. AIDS 2008;22(6):707-15. 14. Friis-Moller N, Reiss P, Sabin CA, et al. Class of antiretroviral drugs and the risk of myocardial infarction. N Engl J Med 2007;356(17):1723-35. 15. Kaplan RC, Kingsley LA, Sharrett AR, et al. Ten-year predicted coronary heart disease risk in HIV-infected men and women. Clin Infect Dis 2007;45(8):1074-81. 16. Triant VA, Lee H, Hadigan C, Grinspoon SK. Increased acute myocardial infarction rates and cardiovascular risk factors among patients with human immunodeficiency virus disease. J Clin Endocrinol Metab 2007;92(7):2506-12. 17. Calza L, Manfredi R, Pocaterra D, Chiodo F. Risk of premature atherosclerosis and ischemic heart disease associated with HIV infection and antiretroviral therapy. J Infect 2008;57(1):16-32. 18. Currier JS, Lundgren JD, Carr A, et al. Epidemiological evidence for cardiovascular disease in HIV-infected patients and relationship to highly active antiretroviral therapy. Circulation 2008;118(2):e29-35. 19. Lang S, Mary-Krause M, Cotte L, et al. Impact of Individual Antiretroviral Drugs on the Risk of Myocardial Infarction in Human Immunodeficiency Virus–Infected Patients: A Case-Control Study Nested Within the French Hospital Database on HIV ANRS Cohort CO4. Arch Intern Med. 2010;170(14):1228-1238. 20. Deeks SG. HIV infection, inflammation, immunosenescence, and aging. Annu Rev Med 2011;62:141-55. 21. Rickabaugh TM, Jamieson BD. A challenge for the future: aging and HIV infection. Immunol Res 2010;48(1-3):59-71. 22. Pearson TA, Mensah GA, Alexander RW, et al. Markers of inflammation and cardiovascular disease: application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 2003;107(3):499-511. 23. Appay V, Sauce D, eds. Handbook on Immunosenescence: Springer Science + Business Media; 2009. 24. General Cardiovascular Disease (10-year Risk). National Heart, Lung, and Blood Institute, 2011. (Accessed February 8, 2011, at http://www.framinghamheartstudy.org/risk/gencardio.html.) 25. Saves M, Chene G, Ducimetiere P, et al. Risk factors for coronary heart disease in patients treated for human immunodeficiency virus infection compared with the general population. Clin Infect Dis 2003;37(2):292-8. 26. Medina S, Wessman D, Krause D, Stepenosky J, Boswell G, Crum-Cianflone N. Coronary aging in HIV-infected patients. Clin Infect Dis 2010;51(8):990-3. 27. Guaraldi G, Zona S, Alexopoulos N, et al. Coronary aging in HIV-infected patients. Clin Infect Dis 2009;49(11):1756-62. 28. Worm SW, De Wit S, Weber R, et al. Diabetes mellitus, preexisting coronary heart disease, and the risk of subsequent coronary heart disease events in patients infected with human immunodeficiency virus: the Data Collection on Adverse Events of Anti-HIV Drugs (D:A:D Study). Circulation 2009;119(6):805-11. 29. Onen NF, Overton ET. A Review of Premature Frailty in HIV-infected Persons; Another Manifestation of HIV-Related Accelerated Aging. Curr Aging Sci 2010. 30. Friis-Moller N, Sabin CA, Weber R, et al. Combination antiretroviral therapy and the risk of myocardial infarction. N Engl J Med 2003;349(21):1993-2003. 31. Sudano I, Spieker LE, Noll G, Corti R, Weber R, Luscher TF. Cardiovascular disease in HIV infection. Am Heart J 2006;151(6):1147-55. 32. Colafigli M, Di Giambenedetto S, Bracciale L, Tamburrini E, Cauda R, De Luca A. Cardiovascular risk score change in HIV-1-infected patients switched to an atazanavir-based combination antiretroviral regimen. HIV Med 2008;9(3):172-9. 33. Sabin CA, Worm SW, Weber R, et al. Use of nucleoside reverse transcriptase inhibitors and risk of myocardial infarction in HIV-infected patients enrolled in the D:A:D study: a multi-cohort collaboration. Lancet 2008;371(9622):1417-26. 34. Costagliola D, Lang S, Mary-Krause M, Boccara F. Abacavir and cardiovascular risk: reviewing the evidence. Curr HIV/AIDS Rep 2010;7(3):127-33. 35. Martinez E, Larrousse M, Podzamczer D, et al. Abacavir-based therapy does not affect biological mechanisms associated with cardiovascular dysfunction. AIDS 2010;24(3):F1-9. 36. Humphries A, Amin J, Cooper D. Changes in cardiovascular biomarkers with abacavir: a randomized 96-week trial. 17th Conference on Retroviruses and Opportunistic Infections 2010;Abstract 718:San Francisco. 37. Age-standardized mortality rates by selected causes, by sex. 2010. (Accessed April 8, 2011, at http://www40.statcan.gc.ca/l01/cst01/health30a-eng.htm.) 38. D’Agostino RB, Sr., Grundy S, Sullivan LM, Wilson P. Validation of the Framingham coronary heart disease prediction scores: results of a multiple ethnic groups investigation. JAMA 2001;286(2):180-7. 39. Conroy RM, Pyorala K, Fitzgerald AP, et al. Estimation of ten-year risk of fatal cardiovascular disease in Europe: the SCORE project. Eur Heart J 2003;24(11):987-1003. 40. Reynolds Risk Score. 2011. (Accessed at http://www.reynoldsriskscore.org/.) 41. Greenland P, Alpert JS, Beller GA, et al. 2010 ACCF/AHA guideline for assessment of cardiovascular risk in asymptomatic adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2010;122(25):e584-636. 42. Mondy KE, Gottdiener J, Overton ET, et al. High Prevalence of Echocardiographic Abnormalities among HIV-infected Persons in the Era of Highly Active Antiretroviral Therapy. Clin Infect Dis 2011;52(3):378-86. 43. Grinspoon SK, Grunfeld C, Kotler DP, et al. State of the science conference: Initiative to decrease cardiovascular risk and increase quality of care for patients living with HIV/AIDS: executive summary. Circulation 2008;118(2):198-210.
Chapter 2: Cardiovascular Disease
Early death from cardiovascular (CV) complications in aging individuals infected with HIV is a growing challenge in chronic HIV management. There are numerous factors that have converged to increase CV risk in individuals infected with HIV compared to age-matched individuals without infection. These include a greater prevalence of CV risk factors, the adverse effects of several antiretroviral agents that appear to contribute to CV risk, and a direct or indirect effect of HIV itself. In HIV patients with otherwise well controlled viremia, CV disease is expected to become an increasingly important cause of preventable death. The effort to modify the impact of this potential crisis depends on heightened awareness of the risks in HIV patients and aggressive monitoring and intervention for modifiable risk factors. Individualization of therapy will be increasingly important when attempting to reduce CV risks in the context of sustained HIV suppression.
Show review